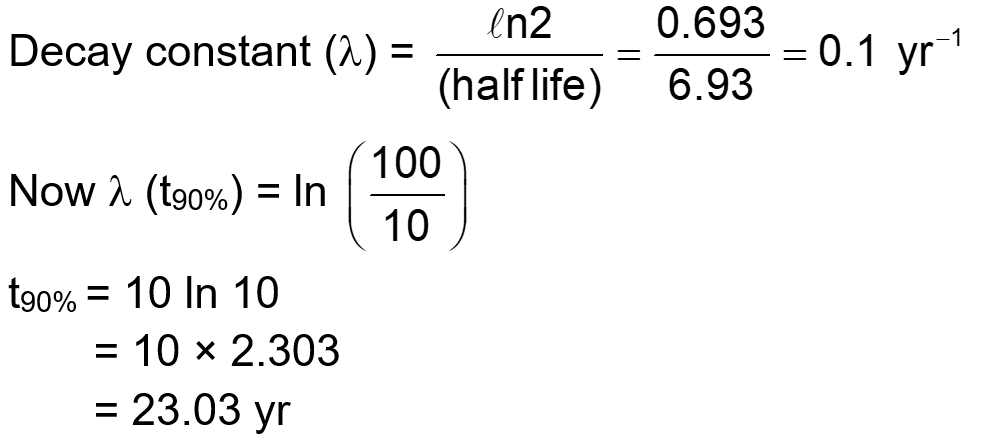During the nuclear explosion, one of the products is ${ }^{90} \mathrm{Sr}$ with half life of 6.93 years. If $1 \mu \mathrm{~g}$ of ${ }^{90} \mathrm{Sr}$ was absorbed in the bones of a newly born baby in place of Ca , how much time, in years, is required to reduce it by $90 \%$ if it is not lost metabolically