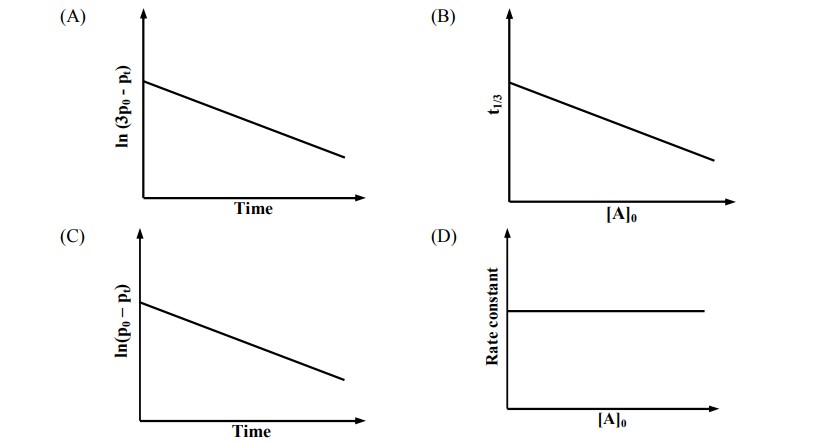
For a first order reaction $\mathrm{A}(\mathrm{g}) \rightarrow 2 \mathrm{~B}(\mathrm{~g})+\mathrm{C}(\mathrm{g})$ at constant volume and 300 K , the total pressure at the beginning ( $t=0$ ) and at time $t$ are $P_0$ and $P_t$, respectively. Initially, only $A$ is present with concentration $[\mathrm{A}]_0$, and $\mathrm{t}_{1 / 3}$ is the time required for the partial pressure of A to reach $1 / 3^{\text {rd }}$ of its initial value. The correct option(s) is (are) (Assume that all these gases behave as ideal gases)