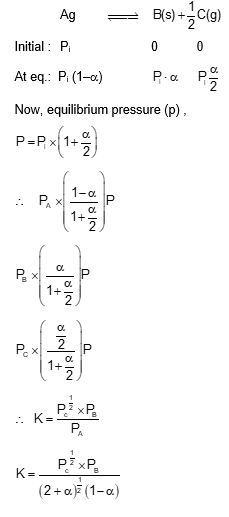
For a reaction at equilibrium
$$
\mathrm{A}(\mathrm{~g}) \rightleftharpoons \mathrm{B}(\mathrm{~g})+\frac{1}{2} \mathrm{C}(\mathrm{~g})
$$
the relation between dissociation constant (K), degree of dissociation ( $\alpha$ ) and equilibrium pressure (p) is given by :