For the cell $\mathrm{Zn}(\mathrm{s})\left|\mathrm{Zn}^{2+}(\mathrm{ag}) \| \mathrm{M}^{x+}(\mathrm{ag})\right| \mathrm{M}(\mathrm{s})$, different half cells and their standard electrode potentials are given below
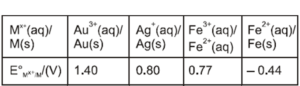
If $\mathrm{E}_{\mathrm{Zn}^{2+} / \mathrm{Zn}}^{\circ}=-0.76 \mathrm{~V}$, which cathode will give a maximum value of $\mathrm{E}_{\text {meal }}^{\circ}$ per electron transferred?
