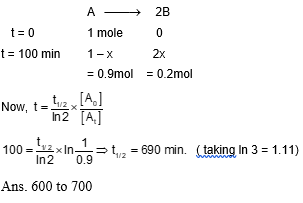
For the first order reaction $A \rightarrow 2 B, 1$ mole of reactlant $A$ gives 0.2 moles of $B$ after 100 minutes. The half life of the reaction is $\_\_\_\_$ min . (Round off to the nearest integer).
[Use : In $2=0.69, \ln 10=2.3$ Properties of logarithms: In $x y=y \ln x ; \ln \left(\frac{x}{y}\right)=\ln x-\ln y$ ]