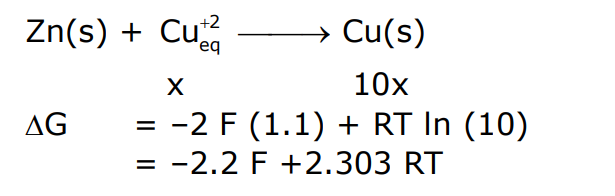For the following cell,
$$
\mathrm{Zn}(\mathrm{~s})\left|\mathrm{ZnSO}_4(\mathrm{aq})\right|\left|\mathrm{CuSO}_4(\mathrm{aq})\right| \mathrm{Cu}(\mathrm{~s})
$$
When the concentration of $\mathrm{Zn}^{2+}$ is 10 times the concentration of $\mathrm{Cu}^{2+}$, the expression for $\Delta \mathrm{G}$ (in J $\mathrm{mol}^{-1}$ ) is
$$
\left[\mathrm{F} \text { is faraday constant; } \mathrm{R} \text { is gas constant; } \mathrm{T} \text { is temperature; } \mathrm{E}^0(\text { cell })=1.1 \mathrm{~V}\right]
$$