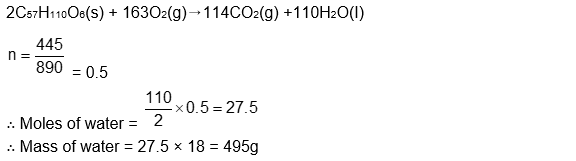For the following reaction, the mass of water produced from 445 g of $\mathrm{C}_{57} \mathrm{H}_{110} \mathrm{O}_6$ is:
$
2 \mathrm{C}_{57} \mathrm{H}_{110} \mathrm{O}_6(\mathrm{~s})+163 \mathrm{O}_2(\mathrm{~g}) \rightarrow 114 \mathrm{CO}_2(\mathrm{~g})+110 \mathrm{H}_2 \mathrm{O}(\mathrm{l})
$