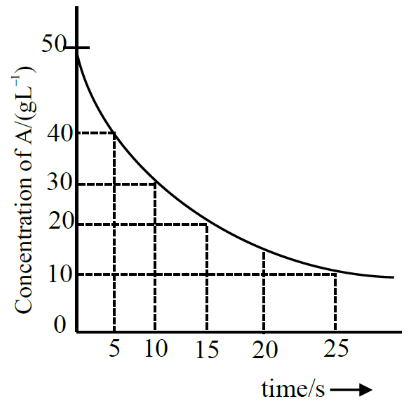
For the reaction $A \rightarrow B$ the following graph was obtained. The time required (in seconds) for the concentration of $A$ to reduce to $2.5 g L^{-1}$ (if the initial concentration of A was $50 g L^{-1}$ ) is (Nearest integer)
Given : $\log 2=0.3010$