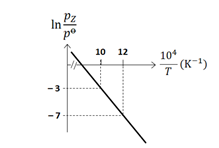
For the reaction, $\mathbf{X}(s) \rightleftharpoons \mathbf{Y}(s)+\mathbf{Z}(g)$, the plot of $\ln \frac{\mathrm{p}_{\mathrm{z}}}{\mathrm{p}^{\Theta}}$ versus $\frac{10^4}{\mathrm{~T}}$ is given below (in solid line), where pz is the pressure (in bar) of the gas Z at temperature T and $\mathrm{p}^{\ominus}=1$ bar.
$$
\text { (Given, } \frac{d(\ln K)}{d\left(\frac{1}{T}\right)}=-\frac{\Delta H^{\ominus}}{R} \text {, where the equilibrium constant, } K=\frac{p_z}{p^{\ominus}} \text { and the gas constant, }
$$
The value of standard enthalpy, $\Delta \mathrm{H}^{\ominus}$ (in $\mathrm{kJ} \mathrm{mol}^{-1}$ ) for the given reaction is $\_\_\_\_$ -

