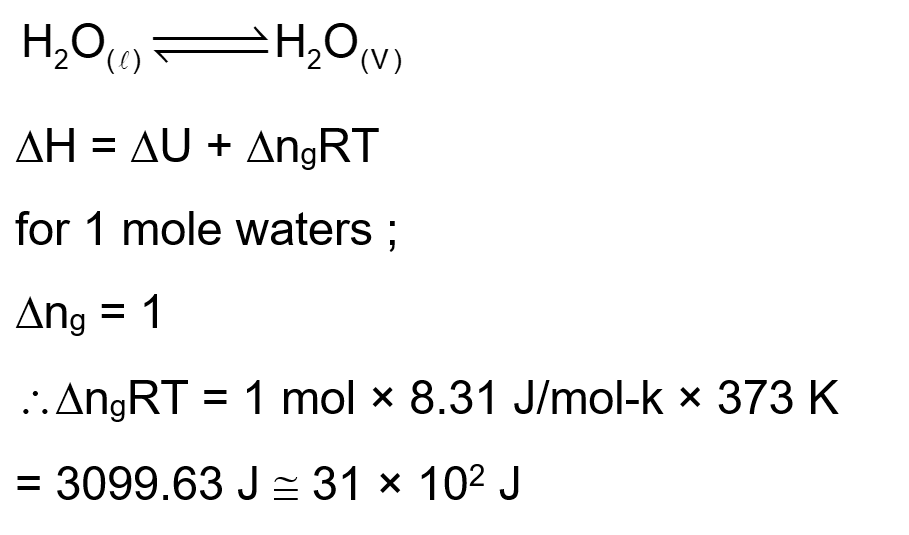For water at $100^{\circ} \mathrm{C}$ and $1 \mathrm{bar}, \Delta_{\text {vap }} \mathrm{H}-\Delta_{\text {vap }} \mathrm{U}=$ $\_\_\_\_$ $\times 10^2 \mathrm{~J} \mathrm{~mol}^{-1}$.
(Round off to the Nearest Integer)
[Use : $\mathrm{R}=8.31 \mathrm{~J} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ ]