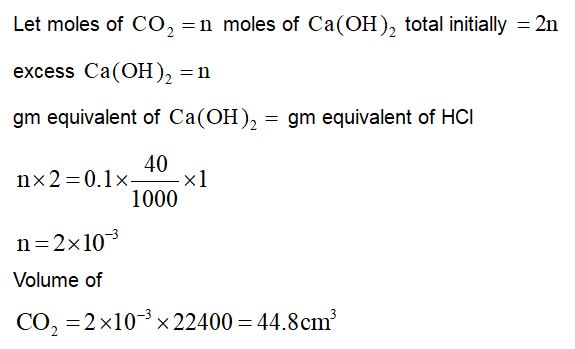Some $\mathrm{CO}_2$ gas was kept in a sealed container at a pressure of 1 atm and at 273 K . This entire amount of $\mathrm{CO}_2$ gas was later passed through an aqueous solution of $\mathrm{Ca}(\mathrm{OH})_2$. The excess unreacted $\mathrm{Ca}(\mathrm{OH})_2$ was later neutralized with 0.1 M of 40 mL HCl . If the volume of the sealed container of $\mathrm{CO}_2$ was $x$, then $x$ is $\_\_\_\_$ $\mathrm{cm}^3$ (nearest integer).