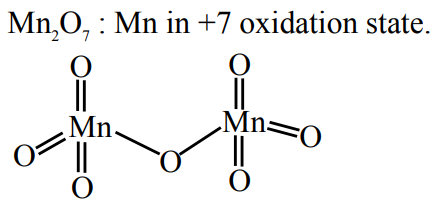
Given below are some of the statements about Mn and $\mathrm{Mn}_2 \mathrm{O}_7$. Identify the correct statements.
A. Mn forms the oxide $\mathrm{Mn}_2 \mathrm{O}_7$, in which Mn is in its highest oxidation state.
B. Oxygen stabilizes the Mn in higher oxidation states by forming multiple bonds with Mn .
C. $\mathrm{Mn}_2 \mathrm{O}_7$ is an ionic oxide.
D. The structure of $\mathrm{Mn}_2 \mathrm{O}_7$ consists of one bridged oxygen.
Choose the correct answer from the options given below :