Given below are two statements :
Statement (I) : For $\mathrm{ClF}_3$, all three possible structures may be drawn as follows.
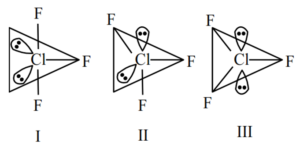
Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the lp - bp repulsion is minimum.
In the light of the above statements, choose the most appropriate answer from the options given below :
 Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the lp - bp repulsion is minimum.
Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the lp - bp repulsion is minimum.