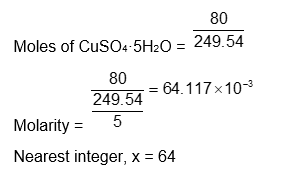If 80 g of copper sulphate $\mathrm{CuSO}_4 \cdot 5 \mathrm{H}_2 \mathrm{O}$ is dissolved in deionised water to make 5 L of solution. The concentration of the copper sulphate solution is $\mathrm{x} \times 10^{-3}$ mol $\mathrm{L}^{-1}$. The value of $x$ is $\_\_\_\_$ .
[Atomic masses $\mathrm{Cu}: 63.54 \mathrm{u}, \mathrm{S}: 32 \mathrm{u}, \mathrm{O}: 16 \mathrm{u}, \mathrm{H}: 1 \mathrm{u}$ ]