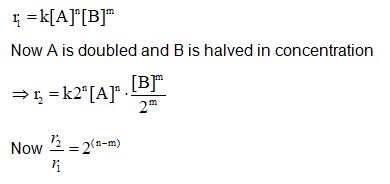Rate law for a reaction between A and B is given by
${\rm{r}} = {\rm{k}}{[\;{\rm{A}}]^{\rm{n}}}{[\;{\rm{B}}]^{\rm{m}}}$
If concentration of A is doubled and concentration of B is halved from their initial value, the ratio of new rate of reaction to the initial rate of reaction $\left( {\frac{{{r_2}}}{{{r_1}}}} \right)$ is