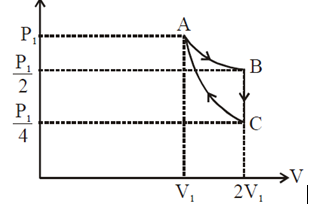
If one mole of an ideal gas at $\left(P_1, V_1\right)$ is allowed to expand reversibly and isothermally (A to B) its pressure is reduced to one-half of the original pressure (see figure). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value $(B \rightarrow C)$. Then it is restored to its initial state by a reversible adiabatic compression ( C to A ). The net workdone by the gas is equal to: