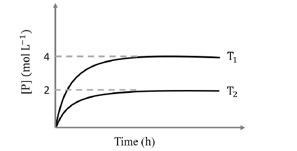
 If $T_1=2 T_2$ and $\left(\Delta G_2^{\ominus}-\Delta G_1^{\ominus}\right)=R T_2 \ln x$, then the value of $x$ is $\_\_\_\_$
[ $\Delta \mathrm{G}_1^{\infty}$ and $\Delta \mathrm{G}_2^{\circ}$ are standard Gibb's free energy change for the reaction at temperatures $\mathrm{T}_1$ and $\mathrm{T}_2$, respectively.]
If $T_1=2 T_2$ and $\left(\Delta G_2^{\ominus}-\Delta G_1^{\ominus}\right)=R T_2 \ln x$, then the value of $x$ is $\_\_\_\_$
[ $\Delta \mathrm{G}_1^{\infty}$ and $\Delta \mathrm{G}_2^{\circ}$ are standard Gibb's free energy change for the reaction at temperatures $\mathrm{T}_1$ and $\mathrm{T}_2$, respectively.]