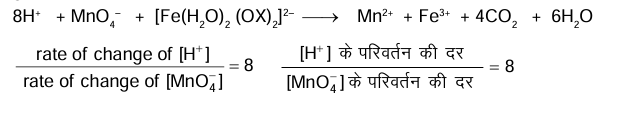
In dilute aqueous $\mathrm{H}_2 \mathrm{SO}_4$, the complex diaquodioxalatoferrate $(\mathrm{II})$ is oxidized by $\mathrm{MnO}_4^{-}$. For this reaction, the ratio of the rate of change of $\left[\mathrm{H}^{+}\right]$to the rate of change of $\left[\mathrm{MnO}_4^{-}\right]$is