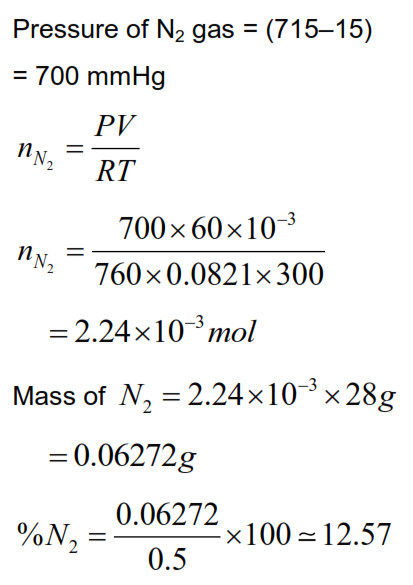In Dumas' method for estimation of nitrogen, 0.5 gram of an organic compound gave 60 mL of nitrogen collected at 300 K temperature and 715 mm Hg pressure. The percentage composition of nitrogen in the compound (Aqueous tension at $300 \mathrm{~K}=15 \mathrm{~mm} \mathrm{Hg}$ ) is $\_\_\_\_$ \%