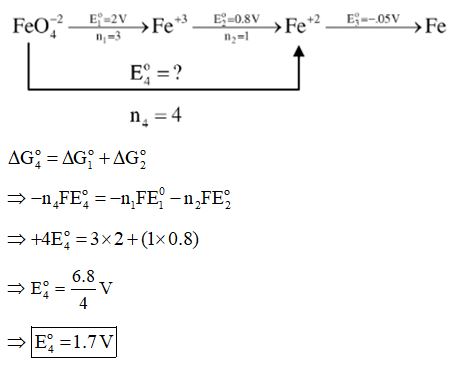
$\text{FeO}_{4}^{{2-}}\xrightarrow{{+2.0~\text{V}}}\text{F}{{\text{e}}^{{3+}}}\xrightarrow{{0.8~\text{V}}}\text{F}{{\text{e}}^{{2+}}}\xrightarrow{{-0.5~\text{V}}}\text{F}{{\text{e}}^{0}}$
In the above diagram, the standard electrode potentials are given in volts (over the arrow). The value of ${{\text{E}}_{{\text{FeO}_{4}^{{2-}}/\text{F}{{\text{e}}^{{2+}}}}}}$ is