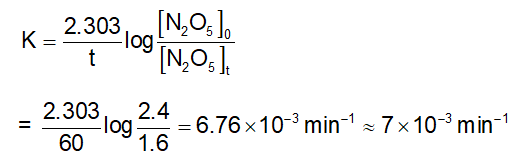In the above first order reaction the initial concentration of $\mathrm{N}_2 \mathrm{O}_5$ is $2.40 \times 10^{-2} \mathrm{~mol} \mathrm{~L}^{-1}$ at 318 K. The concentration of $\mathrm{N}_2 \mathrm{O}_5$ after 1 hour was $1.60 \times 10^{-2} \mathrm{~mol} \mathrm{~L}^{-1}$. The rate constant of the reaction at 318 K is _______ $\times 10^{-3} \mathrm{~min}^{-1}$. (Nearest integer)
[Given : log 3 = 0.477, log 5 = 0.699]