Consider the following transformation involving first order elementary reaction in each step at constant temperature as shown below.
$\mathrm{A}+\mathrm{B} \underset{\text { Step 3 }}{\stackrel{\text { Step 1 }}{\rightleftharpoons}} \mathrm{C} \xrightarrow{\text { Step 2 }} \mathrm{P}$
Some details of the above reaction are listed below.
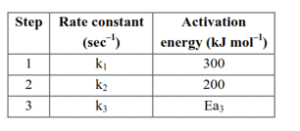
If the overall rate constant of the above transformation ( $k$ ) is given as $k=\frac{k_1 k_2}{k_3}$ and the overall activation energy $\left(E_a\right)$ is $400 \mathrm{~kJ} \mathrm{~mol}^{-1}$, then the value of $\mathrm{Ea}_3$ is $\$$ iqquad $\$ \mathrm{kJmol}^{-1}$ (nearest integer)
 If the overall rate constant of the above transformation ( $k$ ) is given as $k=\frac{k_1 k_2}{k_3}$ and the overall activation energy $\left(E_a\right)$ is $400 \mathrm{~kJ} \mathrm{~mol}^{-1}$, then the value of $\mathrm{Ea}_3$ is $\$$ iqquad $\$ \mathrm{kJmol}^{-1}$ (nearest integer)
If the overall rate constant of the above transformation ( $k$ ) is given as $k=\frac{k_1 k_2}{k_3}$ and the overall activation energy $\left(E_a\right)$ is $400 \mathrm{~kJ} \mathrm{~mol}^{-1}$, then the value of $\mathrm{Ea}_3$ is $\$$ iqquad $\$ \mathrm{kJmol}^{-1}$ (nearest integer)