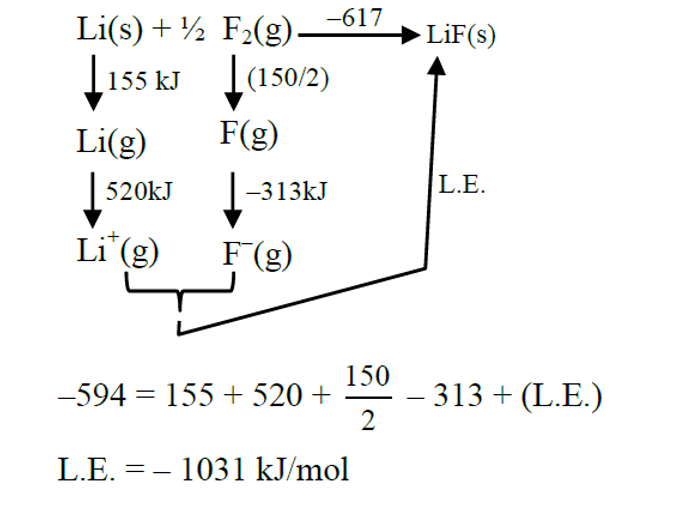
If the enthalpy of sublimation of Li is $155 \mathrm{~kJ} \mathrm{~mol}^{-1}$, enthalpy of dissociation of $\mathrm{F}_2$ is $150 \mathrm{~kJ} \mathrm{~mol}^{-1}$, ionization enthalpy of Li is $520 \mathrm{~kJ} \mathrm{~mol}^{-1}$, electron gain enthalpy of F is $-313 \mathrm{~kJ} \mathrm{~mol}^{-1}$, standard enthalpy of formation of LiF is $-594 \mathrm{~kJ} \mathrm{~mol}^{-1}$. The magnitude of lattice enthalpy of LiF is $\_\_\_\_$ $\mathrm{kJmol}^{-1}$
. (Nearest Integer)