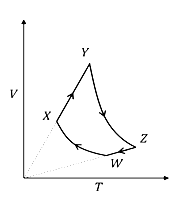
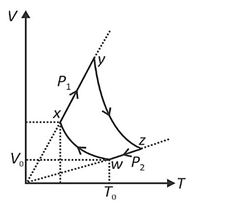
An ideal monatomic gas of n moles is taken through a cycle WXYZW consisting of consecutive adiabatic and isobaric quasi-static processes, as shown in the schematic V-T diagram. The volume of the gas at W, X and Y points are,$64 \mathrm{~cm}^3, 125 \mathrm{~cm}^3$ and $250 \mathrm{~cm}^3$
respectively. If the absolute temperature of the gas $T_W$ at the point $W$ is such that $n B J_\alpha=1 J(R$ is the universal gas constant), then the amount of heat absorbed (in $J)$ by the gas along the path $X Y$ is $\_\_\_\_$