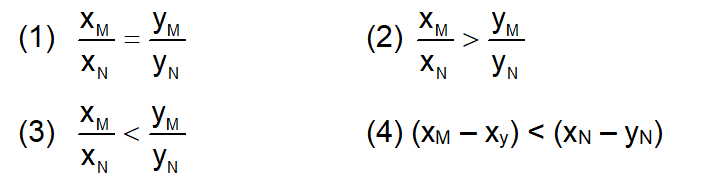Liquid ‘M’ and liquid ‘N’ form an ideal solution. The vapour pressures of pure liquids ‘M’ and ‘N’ are 450 and 700 mmHg, respectively, at the same temperature. Then correct statement is
( $\mathrm{X}_{\mathrm{M}}=$ Mole fraction of ' M ' in solution;
$\mathrm{X}_{\mathrm{N}}=$ Mole fraction of ' N ' in solution;
$\mathrm{y}_{\mathrm{M}}=$ Mole fraction of ' M ' in vapour phase;
$y_N=$ Mole fraction of ' $N$ ' in vapour phase)