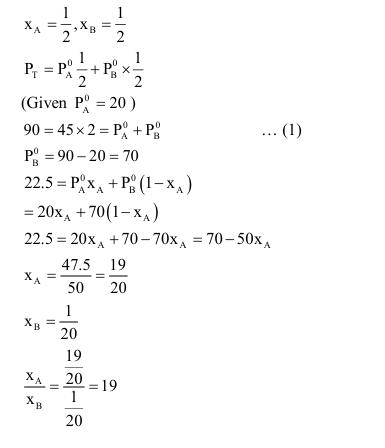Liquids $\mathbf{A}$ and $\mathbf{B}$ form ideal solution over the entire range of composition. At temperature $\mathbf{T}$, equimolar binary solution of liquids $\mathbf{A}$ and $\mathbf{B}$ has vapour pressure 45 Torr. At the same temperature, a new solution of $\mathbf{A}$ and $\mathbf{B}$ having mole fractions $x_{\mathrm{A}}$ and $x_{\mathrm{B}}$, respectively, has vapour pressure of 22.5 Torr. The value of $x_{\mathrm{A}} / x_{\mathrm{B}}$ in the new solution is $\_\_\_\_$ .
(given that the vapour pressure of pure liquid $\mathbf{A}$ is 20 Torr at temperature T )