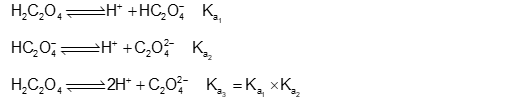$\mathrm{Ka}_1, \mathrm{Ka}_2$ and $\mathrm{Ka}_3$ are the respective ionization constants for the following reactions (a) (b), and (c).
(a) $\mathrm{H}_2 \mathrm{C}_2 \mathrm{O}_4 \rightleftharpoons \mathrm{H}^{+}+\mathrm{HC}_2 \mathrm{O}_4^{-}$ (b) $\mathrm{HC}_2 \mathrm{O}_4^{-} \rightleftharpoons \mathrm{H}^{+}+\mathrm{C}_2 \mathrm{O}_4^{2-}$
(c) $\mathrm{H}_2 \mathrm{C}_2 \mathrm{O}_4 \rightleftharpoons 2 \mathrm{H}^{+}+\mathrm{C}_2 \mathrm{O}_4^{2-}$
The relationship between $K_{a_1}, K_{a_2}$ and $K_{a_3}$ is given as