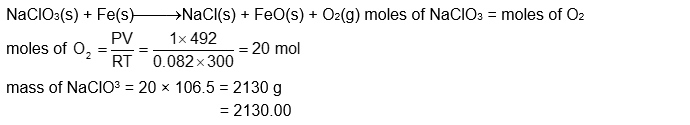$\mathrm{NaClO}_3$ is used, even in spacecrafts, to produce $\mathrm{O}_2$. The daily consumption of pure $\mathrm{O}_2$ by a person is 492 L at $1 \mathrm{~atm}, 300 \mathrm{~K}$. How much amount of $\mathrm{NaClO}_3$, in grams, is required to produce $\mathrm{O}_2$ for the daily consumption of a person at $1 \mathrm{~atm}, 300 \mathrm{~K}$ ? $\_\_\_\_$
$\mathrm{NaClO}_3(\mathrm{~s})+\mathrm{Fe}(\mathrm{s}) \rightarrow \mathrm{O}_2(\mathrm{~g})+\mathrm{NaCl}(\mathrm{s})+\mathrm{FeO}(\mathrm{s})\left(\mathrm{R}=0.082 \mathrm{~L}\right.$ atm $\left.\mathrm{mol}^{-1} \mathrm{~K}^{-1}\right)$