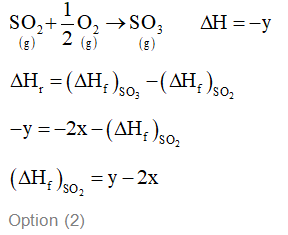$\mathrm{S}(\mathrm{g})+\frac{3}{2} \mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{SO}_{3}(\mathrm{~g})+2 x \mathrm{kcal}$
$\mathrm{SO}_{2}(\mathrm{~g})+\frac{1}{2} \mathrm{O}_{2}(\mathrm{~g}) \rightarrow \mathrm{SO}_{3}(\mathrm{~g})+y \mathrm{kcal}$
The heat of formation of $\mathrm{SO}_{2}(\mathrm{~g})$ is given by :