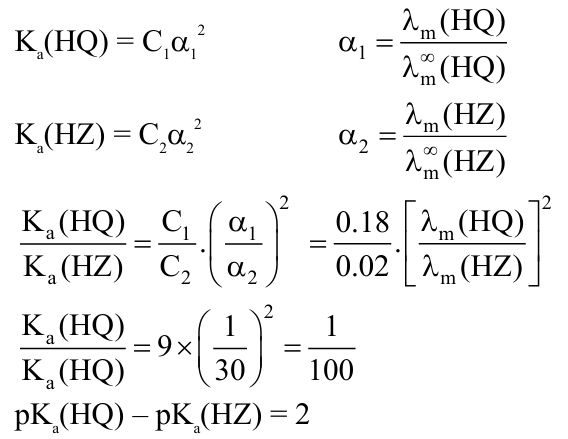Molar conductivity of a weak acid HQ of concentration 0.18 M was found to be $1 / 30$ of the molar conductivity of another weak acid HZ with concentration of 0.02 M . If $\lambda^{\circ} \mathrm{Q}^{-}$happened to be equal with
$\lambda^{\circ} \mathrm{Z}^{-}$, then the difference of the $\mathrm{pK}_{\mathrm{a}}$ values of the two weak acids $\left(\mathrm{pK}_{\mathrm{a}}(\mathrm{HQ})-\mathrm{pK}_{\mathrm{a}}(\mathrm{HZ})\right)$ is - (Nearest integer).
[Given: degree of dissociation $(\alpha) \boxtimes 1$ for both weak acids, $\lambda^{\circ}$ : limiting molar conductivity of ions]