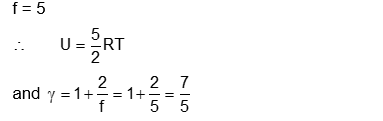Molecules of an ideal gas are known to have three translational degrees of freedom and two rotational degrees of freedom. The gas is maintained at a temperature of T. The total internal energy, $U$ of a mole of this gas, and the value of $\gamma\left(=\frac{C_p}{C_v}\right)$ are given, respectively, by