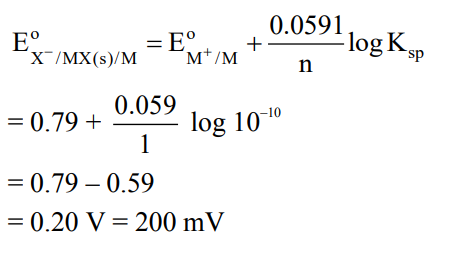MX is a sparingly soluble salt that follows the given solubility equilibrium at 298 K . $\operatorname{MX}(\mathrm{s}) \square \mathrm{M}^{+}(\mathrm{aq})+\mathrm{X}^{-}(\mathrm{aq}) ; \quad \mathrm{K}_{\mathrm{sp}}=10^{-10}$
If the standard reduction potential for $\mathrm{M}^{+}(\mathrm{aq}) \xrightarrow{+\mathrm{e}^{-}} \mathrm{M}(\mathrm{s})$ is $\left(\mathrm{E}_{\mathrm{M}^{+} / \mathrm{M}}^{!}\right)=0.79 \mathrm{~V}$, then the value of the standard reduction potential for the metal/metal insoluble salt electrode $\mathrm{E}_{X^{-} / M X(s) / M}^{!}$is _ $\mathrm{mV} \cdot($ Nearest integer $) \cdot\left[\right.$ Given: $\left.\frac{2.303 \mathrm{RT}}{\mathrm{F}}=0.059 \mathrm{~V}\right]$