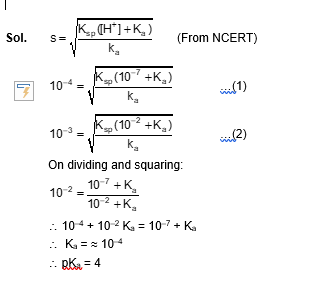
On decreasing the pH from 7 to 2 , the solubility of a sparingly soluble salt $(\mathrm{MX})$ of a weak acid $(\mathrm{HX})$ increased from $10^{-4} \mathrm{~mol} \mathrm{~L}^{-1}$ to $10^{-3} \mathrm{~mol} \mathrm{~L}^{-1}$. The $p \mathrm{~K}_{\mathrm{a}}$ of HX is