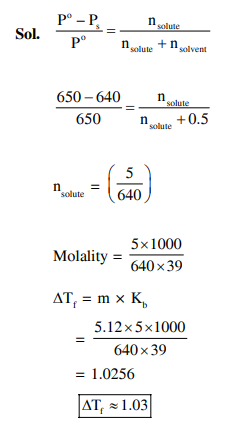On dissolving 0.5 g of a non-volatile non-ionic solute to 39 g of benzene, its vapor pressure decreases from 650 mm Hg to 640 mm Hg . The depression of freezing point of benzene (in K ) upon addition of the solute is $\_\_\_\_$
(Given data : Molar mass and the molal freezing point depression constant of benzene are $78 \mathrm{~g} \mathrm{~mol}^{-1}$ and $5.12 \mathrm{~K} \mathrm{~kg} \mathrm{~mol}^{-1}$, respectively)