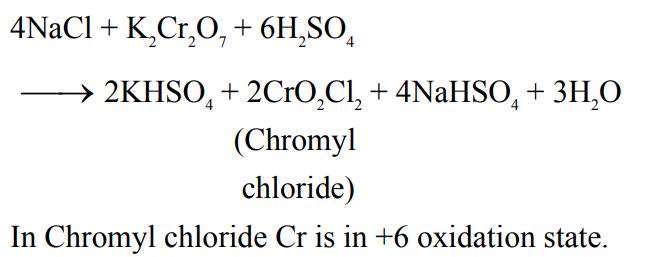On heating a mixture of common salt and $\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7$ in equal amount along with concentrated $\mathrm{H}_2 \mathrm{SO}_4$ in a test tube, a gas is evolved. Formula of the gas evolved and oxidation state of the central metal atom in the gas respectively are: