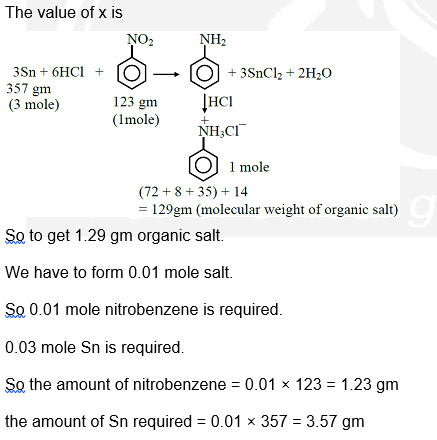
Reaction of $x g$ of Sn with HCl quantitatively produced a salt. Entire amount of the salt reacted with y g of nitrobenzene in the presence of required amount of HCl to produce 1.29 g of an organic salt (quantitatively).
(Use Molar masses (in $\mathrm{g} \mathrm{mol}^{-1}$ ) of H, C, N, O, Cl and Sn as 1, 12, 14, 16, 35 and 119, respectively). The value of $x$ is $\_\_\_\_$ .