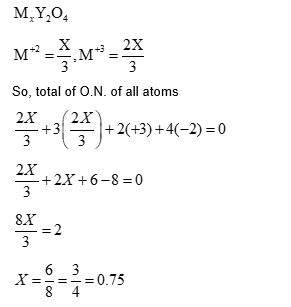In a metal deficient oxide sample, $\mathrm{Mx}_2 \mathrm{Y}_4$ ( M and Y are metals), M is present in both +2 and +3 oxidation states and $\mathbf{Y}$ is in +3 oxidation state. If the fraction of $\mathbf{M}^{2+}$ ions present in $\mathbf{M}$ is $\frac{1}{3}$, the value of $\mathbf{X}$ is