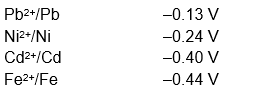 To a solution containing 0.001 M of $\mathrm{X}^{2+}$ and 0.1 M of $\mathrm{Y}^{2+}$, the metal rods X and Y are inserted (at 298 K ) and connected by a conducting wire. This resulted in dissolution of X . The correct combination(s) of $X$ and $Y$, respectively, is(are)
(Given: Gas constant, $\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$,
Faraday constant, $\mathrm{F}=96500 \mathrm{C} \mathrm{mol}^{-1}$ )
To a solution containing 0.001 M of $\mathrm{X}^{2+}$ and 0.1 M of $\mathrm{Y}^{2+}$, the metal rods X and Y are inserted (at 298 K ) and connected by a conducting wire. This resulted in dissolution of X . The correct combination(s) of $X$ and $Y$, respectively, is(are)
(Given: Gas constant, $\mathrm{R}=8.314 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$,
Faraday constant, $\mathrm{F}=96500 \mathrm{C} \mathrm{mol}^{-1}$ )