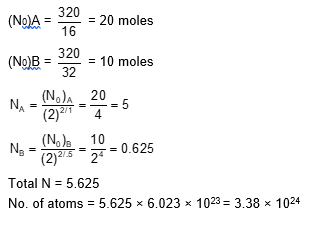Substance A has atomic mass number 16 and half life of 1 day. Another substance B has atomic mass number 32 and half life of $\frac{1}{2}$day. If both A and B simultaneously start undergo radio activity at the same time with initial mass 320 g each, how many total atoms of A and B combined would be left after 2 days.