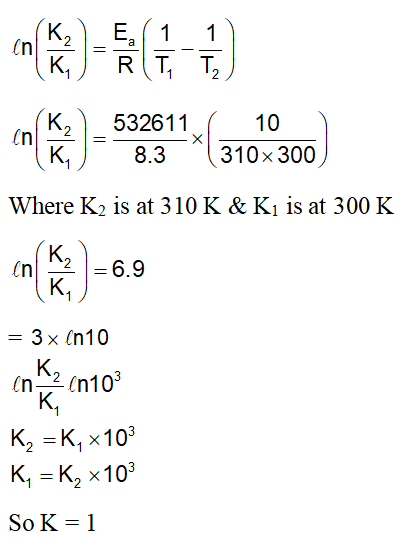The activation energy of one of the reactions in a biochemical process is 532611 J $\mathrm{mol}^{-1}$. When the temperature falls from 310 K to 300 K, the change in rate constant observed is $\mathrm{k}_{300}=\mathrm{x} \times 10^{-3} \mathrm{k}_{310}$. The value of x is _________.
[Given: 1n10 = 2.3
$\left.\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}\right]$