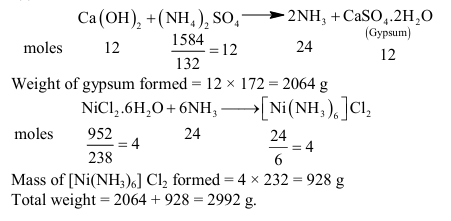
The ammonia prepared by treating ammonium sulphate with calcium hydroxide is completely used by $\mathrm{NiCl}_2 \cdot 6 \mathrm{H}_2 \mathrm{O}$ to form a stable coordination compound. Assume that both the reactions are $100 \%$ complete. If 1584 g of ammonium sulphate and 952 g of $\mathrm{NiCl}_2 \cdot 6 \mathrm{H}_2 \mathrm{O}$ are used in the preparation, the combined weight (in grams) of gypsum and the nickel-ammonia coordination compound thus produced is $\_\_\_\_$ .
(Atomic weights in $\mathrm{g} \mathrm{mol}^{-1}: \mathrm{H}=1, \mathrm{~N}=14, \mathrm{O}=16, \mathrm{~S}=32, \mathrm{Cl}=35.5, \mathrm{Ca}=40, \mathrm{Ni}=59$ )