The amount of energy required to break a bond is same as the amount of energy released when the same bond is formed. In gaseous state, the energy required for homolytic cleavage of a bond is called Bond Dissociation Energy (BDE) or Bond Strength. BDE is affected by scharacter of the bond and the stability of the radicals formed. Shorter bonds are typically stronger bonds. BDEs for some bonds are given below:
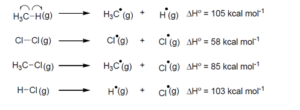
F or the following reaction
$$
\mathrm{CH}_4(\mathrm{~g})+\mathrm{Cl}_2(\mathrm{~g}) \xrightarrow{\text { light }} \mathrm{CH}_3 \mathrm{Cl}(\mathrm{~g})+\mathrm{HCl}(\mathrm{~g})
$$
the correct statement is
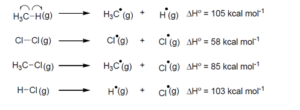 F or the following reaction
$$
\mathrm{CH}_4(\mathrm{~g})+\mathrm{Cl}_2(\mathrm{~g}) \xrightarrow{\text { light }} \mathrm{CH}_3 \mathrm{Cl}(\mathrm{~g})+\mathrm{HCl}(\mathrm{~g})
$$
the correct statement is
F or the following reaction
$$
\mathrm{CH}_4(\mathrm{~g})+\mathrm{Cl}_2(\mathrm{~g}) \xrightarrow{\text { light }} \mathrm{CH}_3 \mathrm{Cl}(\mathrm{~g})+\mathrm{HCl}(\mathrm{~g})
$$
the correct statement is