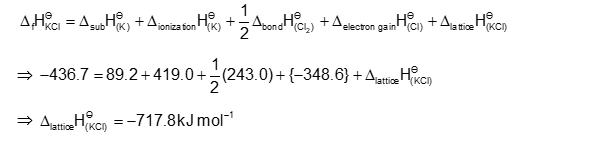
The Born-Haber cycle for KCl is evaluated with the following data:
$
\begin{array}{ll}
\Delta_{\mathrm{f}} \mathrm{H}^{\ominus} \text { for } \mathrm{KCl}=-436.7 \mathrm{~kJ} \mathrm{~mol}^{-1 ;} ; & \Delta_{\text {sub }} \mathrm{H}^{\ominus} \text { for } \mathrm{K}=89.2 \mathrm{~kJ} \mathrm{~mol}^{-1 ;} ; \\
\Delta_{\text {ionization }} \mathrm{H}^{\ominus} \text { for } \mathrm{K}=419 . \mathrm{kJ} \mathrm{~mol}^{-1 ;} ; & \Delta_{\text {electron gain }} \mathrm{H}^{\ominus} \text { for } \mathrm{Cl}_{(\mathrm{g})} \\
=-348.6 \mathrm{~kJ} \mathrm{~mol}^{-1} ; \Delta_{\text {tond }} \mathrm{H}^{\ominus} \text { for } \mathrm{Cl}_2=243 . \mathrm{kJ} \mathrm{~mol}^{-1} &
\end{array}
$
The magnitude of lattice enthalpy of KCl in $\mathrm{kJ} \mathrm{mol}^{-1}$ is $\_\_\_\_$ (Nearest integer)