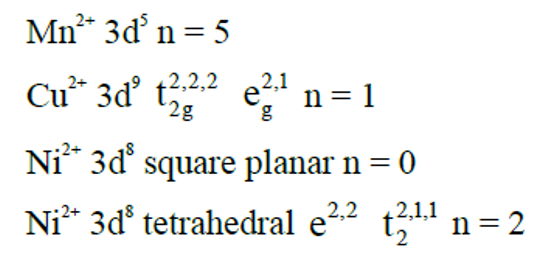The correct increasing order of spin-only magnetic moment values of the complex ions $\left[\mathrm{MnBr}_4\right]^{2-}(\mathrm{A}),\left[\mathrm{Cu}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}(\mathrm{B}),\left[\mathrm{Ni}(\mathrm{CN})_4\right]^{2-}(\mathrm{C})$ and $\left[\mathrm{Ni}\left(\mathrm{H}_2 \mathrm{O}\right)_6\right]^{2+}(\mathrm{D})$ is :