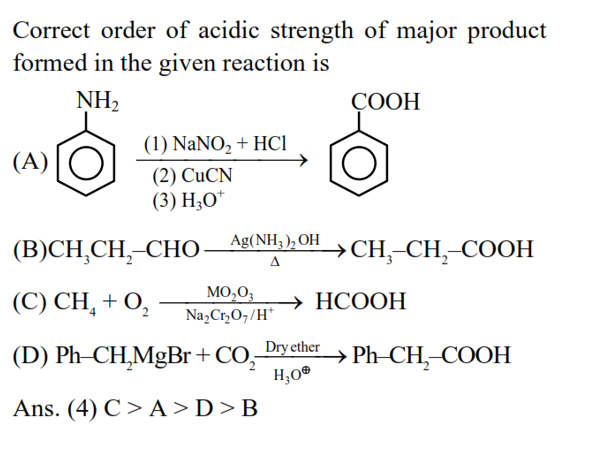
The correct order of acidic strength of the major products formed in the given reactions, is :
A. $\mathrm{PhNH}_2 \xrightarrow[\substack{(2) \mathrm{CuCN} \\(3) \mathrm{H}_3 \mathrm{O}^{+} / \Delta}]{\text { (1) } \mathrm{NaN}_2-\mathrm{HC}\left(\mathrm{C}-\mathrm{S}^{+} \mathrm{C}\right)}[\mathrm{A}]$
B. $\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CHO} \xrightarrow[\Delta]{\left[\mathrm{Ag}\left(\mathrm{NH}_3\right)_3\right]^{+} . \mathrm{OH}^{-}}[\mathrm{B}]$
C. $\mathrm{CH}_4+\mathrm{O}_2 \xrightarrow[\text { (ii) } \mathrm{Na}_2 \mathrm{Cr}_2 \mathrm{O}_7 / \mathrm{H}^{+}]{\text {(a) } \mathrm{Mo}_3 \mathrm{O}_3}[\mathrm{C}]$
D. $\mathrm{PhCH}_2 \mathrm{MgBr}+\mathrm{CO}_2 \xrightarrow[\mathrm{H}_3 \mathrm{O}^{+}]{\text {Dryather }}[\mathrm{D}]$
Choose the correct answer from the options given below :