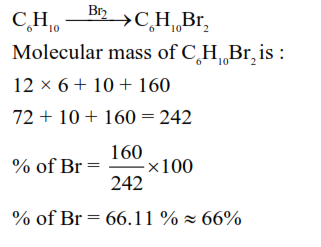The cycloalkene ( $X$ ) on bromination consumes one mole of bromine per mole of ( $X$ ) and gives the product $(\mathrm{Y})$ in which $\mathrm{C}: \mathrm{Br}$ ratio is $3: 1$. The percentage of bromine in the product $(\mathrm{Y})$ is $\_\_\_\_$ $\%$.
(Nearest integer)
(Given : molar mass in $\mathrm{gmol}^{-1} \mathrm{H}: 1, \mathrm{C}: 12, \mathrm{O}: 16, \mathrm{Br}: 80$ )