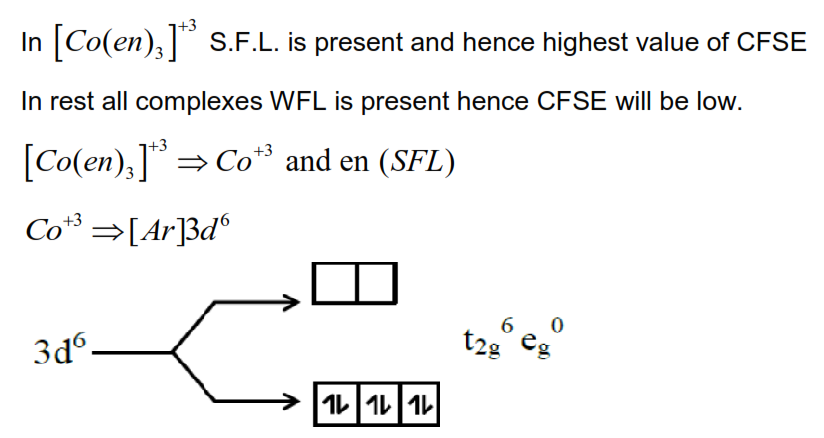
The d-orbital electronic configuration of the complex among $\left[\mathrm{Co}(\mathrm{en})_{3}\right]^{3+},\left[\mathrm{CoF}_{6}\right]^{3-},\left[\mathrm{Mn}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right]^{2+}$ and $\left[\mathrm{Zn}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right]^{2+}$ that has the highest CFSE is :