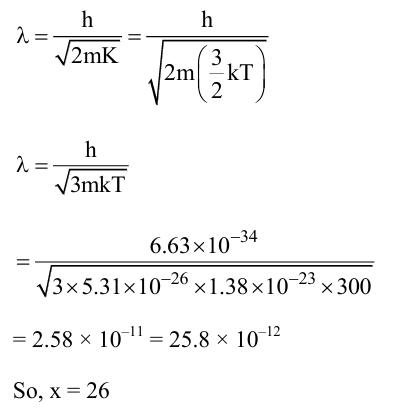The de Broglie wavelength of an oxygen molecule at $27^{\circ} \mathrm{C}$ is $x \times 10^{-12} \mathrm{~m}$. The value of $x$ is (take Planck's constant $=6.63 \times 10^{-34} \mathrm{Js}$, Boltzmann constant $=1.38 \times 10^{-23} \mathrm{~J} / \mathrm{K}$, mass of oxygen molecule $=5.31 \times 10^{-26} \mathrm{~kg}$ )