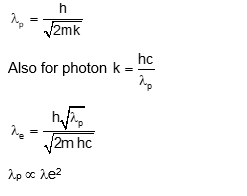
The de Brogue wavelengths for an electron and a photon are $\lambda_{\mathrm{e}}$ and $\lambda_{\mathrm{p}}$ respectively. For the same kinetic energy of electron and photon. which of the following presents the correct relation between the de Brogue wavelengths of two ?