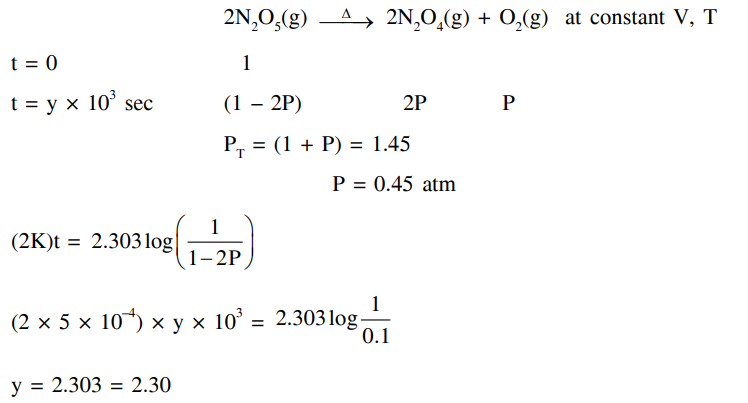The decomposition reaction $2 \mathrm{~N}_2 \mathrm{O}_5(\mathrm{~g}) \xrightarrow{\Delta} 2 \mathrm{~N}_2 \mathrm{O}_4(\mathrm{~g})+\mathrm{O}_2(\mathrm{~g})$ is started in a closed cylinder under isothermal isochoric condition at an initial pressure of 1 atm . After $\mathrm{Y} \times 10^3 \mathrm{~s}$, the pressure inside the cylinder is found to be 1.45 atm . If the rate constant of the reaction is $5 \times 10^{-4} \mathrm{~s}^{-1}$, assuming ideal gas behavior, the value of Y is $\_\_\_\_$